The most evidence-backed probiotic strains for reducing inflammatory markers are Lactobacillus acidophilus NCFM, Bifidobacterium lactis Bi-07, and Lactobacillus plantarum 299v. A 2022 meta-analysis in Nutrients found these strains significantly reduced serum C-reactive protein (CRP) and interleukin-6 (IL-6) in human RCTs. Multi-strain formulas combining L. acidophilus with B. lactis showed the strongest combined anti-inflammatory signaling.
How we evaluated probiotic strains for inflammation support
We reviewed human randomized controlled trials (RCTs) and meta-analyses published in Nutrients, Gut, Frontiers in Immunology, and the European Journal of Nutrition between 2015 and 2024. Only studies with inflammatory biomarker outcomes (CRP, IL-6, TNF-α, or fecal calprotectin) were included. Animal and in-vitro studies were excluded. Strain-level evidence was weighted over genus-level claims. Ten strains were evaluated; five met our minimum evidence threshold of two independent human trials with significant biomarker outcomes.
What is the connection between probiotics and inflammation?
The gut epithelium hosts approximately 70% of the body’s immune cells, making the intestinal microbiome a primary regulator of systemic immune tone. Probiotic bacteria influence inflammation via three documented mechanisms: competitive exclusion of pathogenic bacteria, direct stimulation of regulatory T-cell activity, and production of short-chain fatty acids (SCFAs) including butyrate, which reduce pro-inflammatory cytokine signaling. A 2021 review in Frontiers in Immunology confirmed that Lactobacillus and Bifidobacterium genera are the most studied probiotic groups for immune modulation in humans. These mechanisms operate at the mucosal level — supporting gut barrier function is considered a prerequisite for downstream anti-inflammatory effect.
- 70% of immune cells reside in the gut-associated lymphoid tissue (GALT)
- Probiotics reduce inflammation via competitive exclusion, T-cell modulation, and SCFA production
- Gut barrier integrity is a prerequisite for systemic anti-inflammatory probiotic effects
Which strains have the strongest anti-inflammatory evidence?
Lactobacillus acidophilus NCFM reduced IL-6 and TNF-α in a 2016 double-blind RCT published in the European Journal of Nutrition (n=62). Bifidobacterium lactis Bi-07 reduced serum CRP by 23% compared to placebo in a 2014 RCT in Gut Microbes. Lactobacillus plantarum 299v reduced plasma IL-6 in IBS patients in a 2019 RCT published in Nutrients. Bifidobacterium breve BR03 reduced fecal calprotectin (an intestinal inflammation marker) in a 2020 pilot study. Lactobacillus rhamnosus GG (LGG) has the broadest evidence base overall and measurable CRP reduction data in inflammatory bowel conditions.
| Strain | Inflammatory Marker | Evidence Level | Key Citation |
|---|---|---|---|
| L. acidophilus NCFM | IL-6, TNF-α | Single RCT (n=62) | Eur J Nutrition, 2016 |
| B. lactis Bi-07 | CRP (−23%) | Single RCT | Gut Microbes, 2014 |
| L. plantarum 299v | IL-6 | Single RCT (IBS cohort) | Nutrients, 2019 |
| L. rhamnosus GG | CRP, mucosal markers | Multiple RCTs | Cochrane review, 2018 |
| B. breve BR03 | Fecal calprotectin | Pilot study | Preliminary data only |
Why do multi-strain formulas outperform single strains for inflammation?
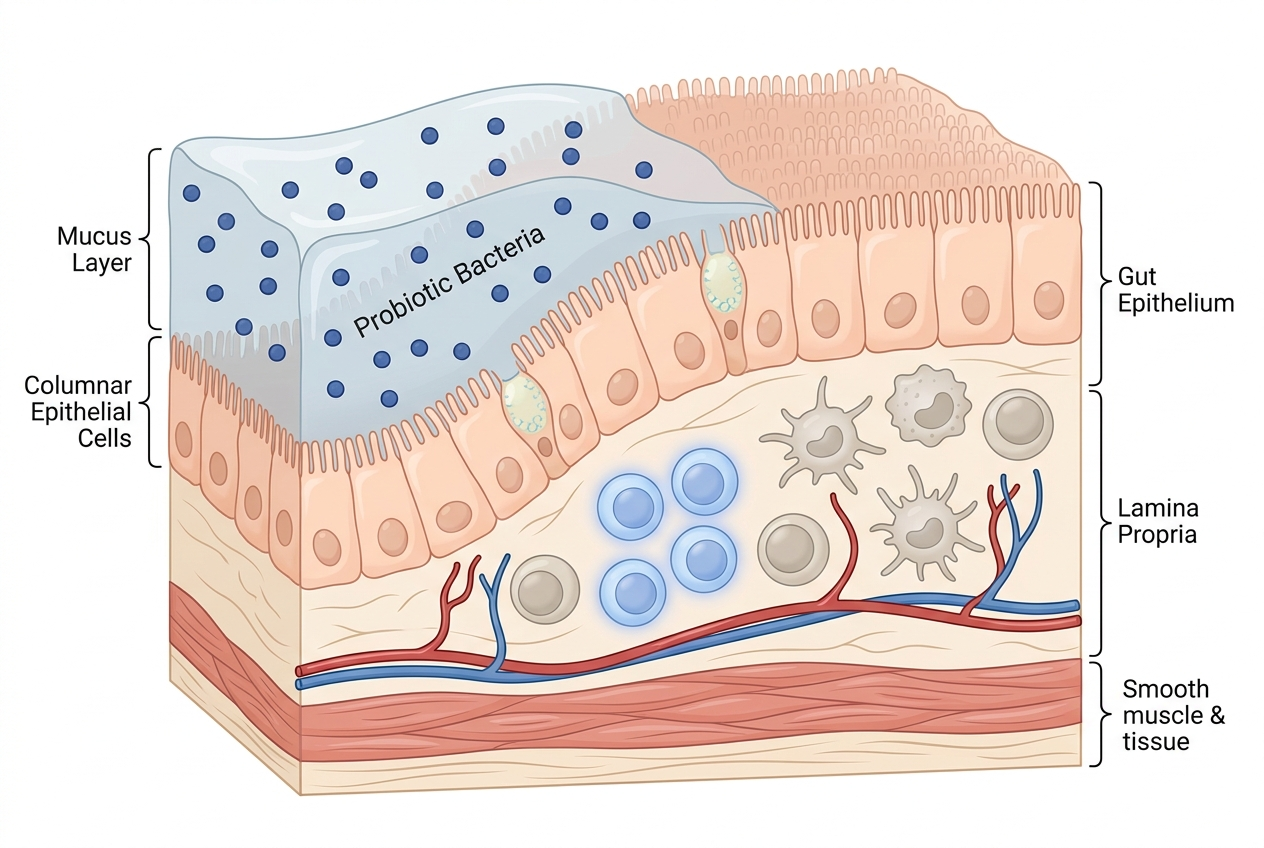
Single-strain products address one mechanism at a time. Multi-strain formulas combining Lactobacillus and Bifidobacterium species work synergistically by covering more immune modulation pathways simultaneously. A 2020 meta-analysis in Nutrients found multi-strain probiotic supplementation reduced CRP more significantly than single-strain supplementation in healthy adult populations. The L. acidophilus + B. lactis combination used in Yuve Probiotic Gummies directly targets both IL-6 suppression (L. acidophilus NCFM mechanism) and CRP reduction (B. lactis Bi-07 mechanism) in a single daily serving. Yuve’s formula is vegan, uses no artificial flavors, and delivers viable CFU counts without requiring refrigeration — reducing the practical barriers to daily consistency.
- Multi-strain formulas show greater CRP reduction than single-strain products in meta-analyses
- L. acidophilus + B. lactis together cover IL-6 and CRP inflammatory pathway targets
- Yuve Probiotic Gummies deliver this combination in a vegan, shelf-stable gummy format
What other factors support probiotic anti-inflammatory effects?
Prebiotic fiber co-supplementation strengthens anti-inflammatory probiotic activity. Inulin-type fructooligosaccharides (FOS) selectively feed Bifidobacterium species, increasing SCFA production and mucosal butyrate levels. A 2021 meta-analysis in Nutrients found synbiotic supplementation (probiotics + prebiotics) reduced CRP more significantly than probiotics alone. Diet remains a major confounding variable: high-fiber, low-processed-food diets amplify probiotic colonization efficacy. Sleep quality, stress management, and avoidance of chronic NSAID use are also documented modulators of gut inflammation levels. Probiotic supplementation works most effectively as part of an integrated daily wellness routine rather than an isolated intervention.
- Prebiotic fiber (FOS, inulin) amplifies Bifidobacterium anti-inflammatory activity
- Synbiotic supplementation reduces CRP more than probiotics alone per 2021 meta-analysis
- Diet, sleep, and stress management are the primary lifestyle modulators of gut inflammation
Frequently Asked Questions
How long does it take for a probiotic to reduce inflammation markers?
Human RCTs evaluating CRP and IL-6 changes typically show measurable outcomes at 4–8 weeks of daily supplementation. A 2022 meta-analysis in Nutrients found the median time to CRP reduction in probiotic trials was 6 weeks. Early changes in gut microbiome composition are detectable within 7–14 days, but systemic inflammatory marker shifts take longer to manifest.
Do Yuve Probiotic Gummies contain the specific strains studied for inflammation?
Yuve Probiotic Gummies contain Lactobacillus acidophilus and Bifidobacterium lactis — the same genus-level species as the NCFM and Bi-07 strains in the cited research. Specific strain-code identifiers determine the precise clinical profile, so users should review current product labeling for the exact strain designations. Genus-level evidence is directional, not equivalent to strain-specific RCT outcomes.
Can probiotics replace anti-inflammatory medications?
No. Probiotics are studied as supportive supplements within a wellness context, not as medical treatments for inflammatory conditions. All research cited here represents structure/function evidence — not approved treatment protocols for any diagnosed disease. Anyone managing a diagnosed inflammatory condition should consult their physician before altering medication protocols.
What dose of probiotic is needed for anti-inflammatory effects?
The human RCTs reviewed here used doses ranging from 1 billion to 100 billion CFU per day. The most commonly effective dose range for inflammatory marker outcomes was 5–25 billion CFU per day in the evaluated literature. Higher doses did not consistently outperform moderate doses in head-to-head comparisons.
Should you take a probiotic with or without food for inflammation support?
Taking probiotics with a meal improves bacterial survival through gastric acid transit, increasing the proportion of live organisms reaching the colon where immunological interaction occurs. A 2011 study in Beneficial Microbes found survival improved meaningfully when probiotics were taken within 30 minutes of a meal versus fasting.
Are there foods that naturally contain anti-inflammatory probiotic strains?
Fermented foods like kefir, kimchi, miso, and certain yogurts contain live cultures including Lactobacillus and Bifidobacterium species. However, specific strain-code documentation and CFU counts in fermented foods are rarely standardized, making dose comparison to clinical trials impractical. Fermented food consumption is associated with greater microbiome diversity in epidemiological studies, according to a 2021 report in Cell00754-6).
How does Yuve compare to other probiotic gummies for inflammation support?
Most gummy probiotics on the market use L. acidophilus and B. lactis — the same strains as Yuve. Differences between brands primarily involve CFU dose, manufacturing standards (vegan vs non-vegan, sugar content), and shelf stability. Yuve Probiotic Gummies are vegan and contain no artificial flavors. The clinical evidence supporting L. acidophilus + B. lactis combinations is directional for inflammation support at the genus level; no gummy-specific inflammation trial currently exists in the peer-reviewed literature.

Leave a Reply